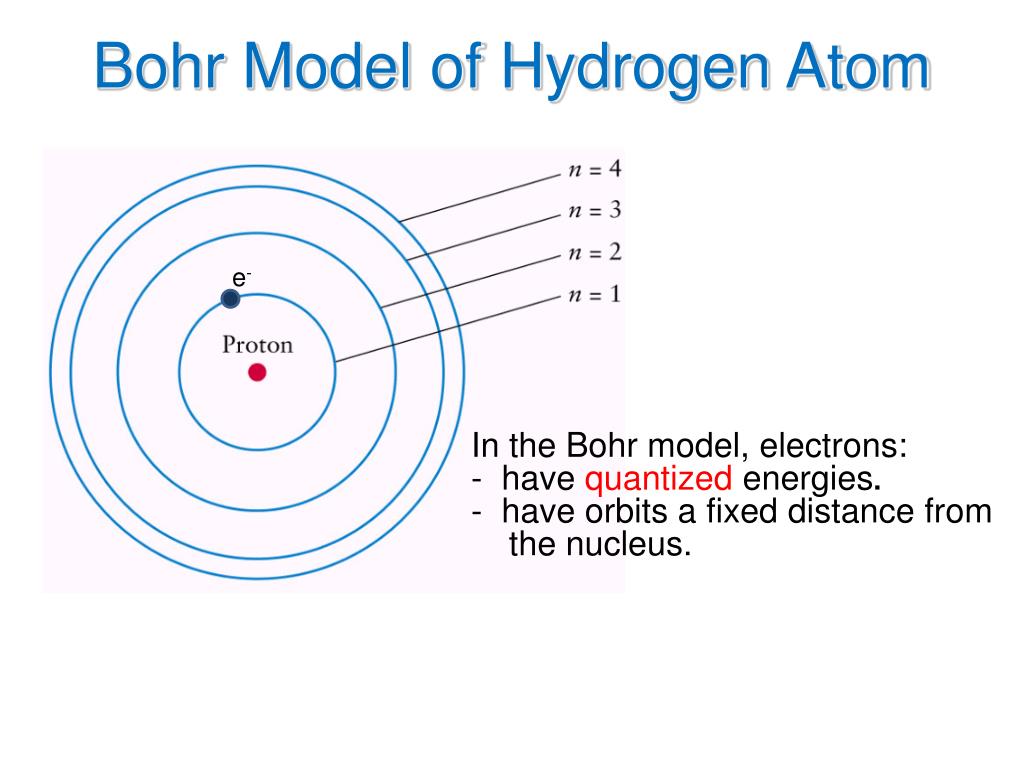
 Total Energy of Electron in Bohr’s Stationary Orbits Since ε 0, h, and e are constants and for a hydrogen atom, Z = 1, r n α (1/n) The velocity of Electron in Bohr’s Stationary Orbits Since ε 0, h, m, e, and p are constants and for a hydrogen atom, Z = 1, r n α n 2 (Ei is the energy of the initial state and Ef is the energy of the final state. Hence, the frequency of the emitted photon is: 
In doing so, a photon is emitted whose energy is equal to the energy difference between the two states. According to this postulate, an electron can transition from a non-radiating orbit to another of a lower energy level. In this postulate, Bohr incorporated early quantum concepts into the atomic theory.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
